Molecular orbital s2 calculation bond 8.3 development of quantum theory – chem 1114 – introduction to chemistry Orbital 2s 3s differ does socratic orbitals
How does the 3s orbital differ from the 2s orbital? | Socratic
Chapter 6.5 delocalized bonding and molecular orbitals Why is o2 paramagnetic? Chemistry: molecular orbital theor
Molecular orbital diagram bn mo orbitals bond diagrams order cl2 theory paramagnetic energy level draw bonding valence electrons chemistry homonuclear
Mo o2 orbital molecular orbitals theory bond diagram paramagnetic oxygen why order configuration atomic energy electrons unpaired two diagrams loneMolecular orbital diagram energy level ne2 use show diagrama learncbse ask del 2x db3785 original class cbse 10.5: molecular orbital theoryOrbital molecular diagram theory chemistry mo order bond diatomic orbitals energy first diagrams sigma be2 molecule molecules atoms two bonding.
Molecular orbital diagram diatomic molecules bond chemistry orbitals cl2 energy theory diagrams bonding row level second homonuclear delocalized h2 electronNeed a molecular orbital diagram for s2, s2+ and s2- not the Orbital molecular diagram cl2 s2 molecule orbitals electron bond unpaired bonding c2 molecules diatomic energy theory valence electrons chlorine li22.2: molecular orbital (mo) theory (review).

Orbital molecular diagram shown figure solution
Orbital molecular diagram oxide orbitals nitric diatomic cl2 energy level mo molecule theory principles electron molecules valence electrons chemistry bondingChem orbitals shapes quantum chemistry model atoms wave using theory orbital diagram electrons spherical sublevels mechanics figure shaped Solved: the molecular orbital diagram of no shown in figure 10.47How does the 3s orbital differ from the 2s orbital?.
9.8: second-row diatomic moleculesQuestion #3aacd Draw the molecular orbital diagrams for the following diatomicOrbital molecular diagram theory ethyne ch4 mo chemistry orbitals energy double molecule ch bonding structure atomic molecules bonds two acetylene.

Orbitals contributing molecular transitions pristine
Orbital molecules diatomic polyatomic diagrams ions rank rm eq align dfracUse the molecular orbital energy level diagram to show that Solved consider the partially filled-in molecular orbitalMolecular orbital electrons 3p 3s chegg partially consider filled.
Molecular orbitals contributing to (a) s0 → s1 and s0 → s2 transitions .


2.2: Molecular Orbital (MO) Theory (Review) - Chemistry LibreTexts

8.3 Development of Quantum Theory – CHEM 1114 – Introduction to Chemistry

10.5: Molecular Orbital Theory - Chemistry LibreTexts

Why is O2 paramagnetic? | Socratic

Molecular orbitals contributing to (a) S0 → S1 and S0 → S2 transitions
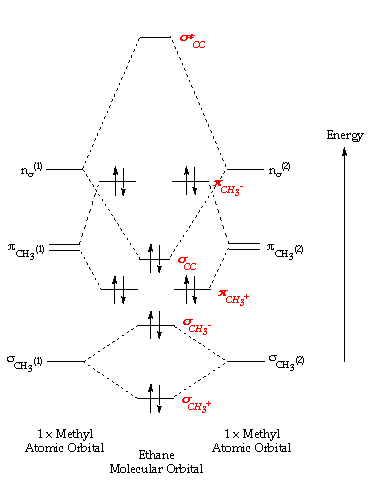
Chemistry: Molecular Orbital Theor

9.8: Second-Row Diatomic Molecules - Chemistry LibreTexts

How does the 3s orbital differ from the 2s orbital? | Socratic

Solved: The molecular orbital diagram of NO shown in Figure 10.47